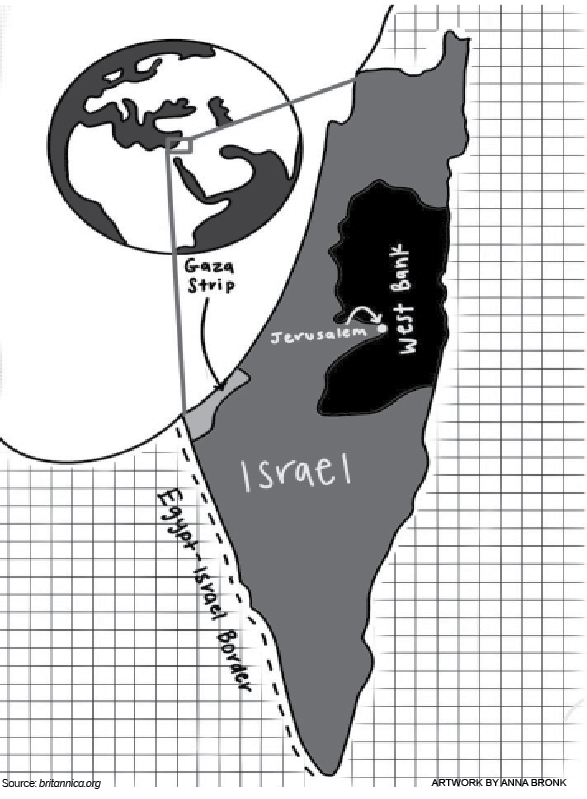
On May 12, research-based biopharmaceutical company Gilead Sciences announced that a new drug called Remdesivir will be given to severe COVID-19 patients to shorten their recovery time. The Food and Drug Administration has authorized the distribution of this drug to certain patients under their Emergency Use Authorization.
Gilead Sciences has been researching Remdesivir for over a decade on fever viruses like Ebola and applied the knowledge from ongoing research of other coronaviruses to the treatment for the SARS-CoV-2 coronavirus when it initially hit. They have been running clinical trials and increasing production of remdesivir since January 2020.
“Patients with COVID-19 who received Remdesivir recovered faster than similar patients who received placebo,” according to Daniel O’Day, Chairman and CEO of Gilead, in an open letter. “There is still more work to do and Remdesivir has not been approved, but all of us at Gilead are humbled by what these promising results might mean for patients.”
The National Institute of Allergies and Infectious Diseases (NIAID) ran a set of clinical trials designed by Gilead to figure out the effect of remdesivir on patients. It was a global, placebo-controlled trial run, which meant that the sample size of patients was from all around the globe, and the patients did not know if they were being given Remdesivir or a sugar pill known as a placebo. This trial run was positive showing that the patients given Remdesivir had a shorter recovery time as opposed to the ones who took the placebo.
“The question of duration is important because the possibility of a shorter treatment course is beneficial in many respects,” said O’Day in his open letter. “Patients can return home earlier from the hospital, families can be reunited, healthcare resources are freed up and more medicine is available for other patients in need.”
The same question of duration was asked when Gilead ran its own clinical trials called SIMPLE trials which had three phases. Each phase conducted the trials on an increased number of hospitalized patients all over the globe. They have 180 trial sites around the world in various countries.
Phase three of the SIMPLE trials tested whether five-day treatments of Remdesivir had similar clinical results to those who received a 10-day treatment. Once the results came back on April 29 saying that both groups of patients had similar clinical improvements, it was concluded that the drug was just as efficient in five days as it was in 10 days, indicating the reduced recovery period in patients.
“The study demonstrates the potential for some patients to be treated with a 5-day regimen, which could significantly expand the number of patients who could be treated with our current supply of Remdesivir,” said Gilead in a press release on April 29. “This is particularly important in the setting of a pandemic, to help hospitals and healthcare workers treat more patients in urgent need of care.”
The FDA has authorized this drug with their EUA because of a public health emergency; however, they have not yet approved Remdesivir for any use outside the scope of the EUA since its safety and efficiency have not been established yet in regard to COVID-19.
“Remdesivir may be effective in treating COVID-19, and that, given there are no adequate, approved, or available alternative treatments, the known and potential benefits to treat this serious or life-threatening virus currently outweigh the known and potential risks of the drug’s use,” according to an FDA press release.
Because of the EUA, patients, caregivers and healthcare providers have been given a fact sheet detailing the effects of Remdesivir and how to administer the drug. It has sections explaining COVID-19, the symptoms of the virus, important reminders for patients, other possible treatment options, the side effects of remdesivir and potential consequences of not taking the drug.
“There’s tremendous interest among all parties to identify and arm ourselves with medicines to combat COVID-19, and through our Coronavirus Treatment Acceleration Program, the FDA is working around-the-clock and using every tool at our disposal to speed these efforts,” said FDA Commissioner Stephen M. Hahn, M.D. in a news release.
Gilead Sciences has been speeding up production of the drug since January while still working within all the necessary precautions that need to be taken during this complex process.
“Our existing supply, including finished product ready for distribution as well as materials in the final stages of production, amounts to 1.5 million individual doses,” said O’Day in the open letter. “We had estimated that this would be 140,000 treatment courses based on a 10-day treatment duration.”
In an announcement by President Trump on May 1, Trump invited O’Day to give a statement on the effectiveness of remdesivir and the way it can combat the coronavirus. O’Day brought up that the drug is the first step to combatting the virus and that it is still the early stages of the coronavirus itself.
“With this base step, with an antiviral like remdesivir, that the way to actually even get better results is to add medicines on top of an antiviral,” said O’Day in the White House remarks issued on May 1. “That was really how we were able to get HIV to a chronic illness: by a combination of therapies.”
Upon hearing about the drug, the president had high hopes for its effectiveness and ability to treat severe patients after talking with Dr. Hahn, Director of NIAID, Dr. Anthony Fauci and Dr. Deborah Birx, a member of the coronavirus taskforce. He supported the trials run by Gilead and NIAID and commended their efforts in finding this treatment.
“[It’s] an important treatment for hospitalized coronavirus patients … making a contribution to people that are not doing well, people that are sick, people that have this horrible plague that’s set into our country and that we’re getting rid of,” said Trump in the White House’s news release. “It’s really a very promising situation.”